COVID-19 Lung Damage
COVID-19 Lung Damage – Can Inhaled FGF-1 be a Viable Treatment?
It is estimated that approximately 30% of individuals who become infected with the coronavirus and have symptoms will sustain long-term lung damage.
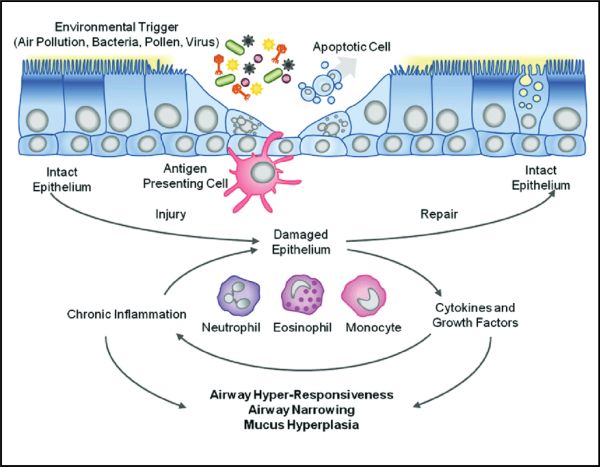
The cells in the lungs which are attacked by the SARS-CoV-2 virus are the epithelial cells. These cells line the airs sacs in the lung and are directly responsible for the proper exchange of oxygen and carbon dioxide in the healthy lung. When epithelial cells become damaged either by the virus directly, or through a hyper-response by the body’s immune system, pneumonia and respiratory distress result. If the epithelial cells cannot be regenerated, long-term lung damage and breathing difficulties persist.
Lung Damage Following COVID-19
Respiratory viral infections are linked to chronic lung diseases, as has been long appreciated following annual influenza epidemics. There is little doubt the same will also hold true with COVID-19 survivors. Early publications indicate that over 80% of patients that had to be hospitalized for COVID-19 had visible lung damage on their CT scan taken at the time of their discharge. After a serious case of COVID-19, it is estimated a patient’s lungs will take 3-12 months to recover to their pre-COVID-19 levels, depending on how much scarring of lung tissue has occurred.
Conclusion
Worldwide, the fifth leading cause of death is from respiratory disorders and a large majority of the individuals dying from these disorders will have lung damage. The emergence and rapid spread of the SARS-CoV-2 virus has dramatically raised the awareness of respiratory distress and lung damage.
The principal cell in the lung that is damaged by respiratory disorders, including COVID-19, are the epithelial cells that line the air sacs of the lung. With epithelial cell damage and destruction, breathing difficulties ensue, and this same process is reproduced in numerous respiratory disorders, whether it be the acute respiratory distress syndrome (ARDS) that is seen in COVID-19 and influenza infections or in emphysema or COPD.
The FGF family of proteins is intimately involved in embryonic lung formation. A number of the FGFs, including FGF-1, are potent stimulators of the proliferation and maturation of lung epithelial cells and have shown excellent efficacy in animal models of lung injury.
Research Papers and additional reading:
- Therapeutic and Pathological Roles of Fibroblast Growth Factors in Pulmonary Diseases. Agha E, Seeger W and Bellusci S. Dev Dynamics 2017; 246:235–244. https://pubmed.ncbi.nlm.nih.gov/27783451/
- The FGF family: biology, pathophysiology and therapy. Beenken A and Mohammadi M. Nat Rev Drug Discov 2009; 8:235-253. https://www.nature.com/articles/nrd2792
- Fibroblast Growth Factor 2 Is Required for Epithelial Recovery, but Not for Pulmonary Fibrosis, in Response to Bleomycin. Guzy D, Ivan Stoilov I, et.al. Amer J of Respir Cell and Mol Biol 2015; 52:116-128. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4370255/
- Fibroblast Growth Factor-1 Therapy for Advanced Emphysema—A New Tissue Engineering Approach for Achieving Lung Volume Reduction. Ingenito E, Tsai L, et. al. J Bronchol. 2006; 13:114–123. https://journals.lww.com/bronchology/Fulltext/2006/07000/Fibroblast_Growth_Factor_1_Therapy_for_Advanced.4.aspx
- Protective effects of basic fibroblast growth factor in the development of emphysema induced by interferon-γ. Lee B-Y, Moon H-G, et.al. Exp Mol Med 2011; 43: 169-178. https://pubmed.ncbi.nlm.nih.gov/21297377/
- Pathogenesis of COVID-19 from a cell biologic perspective. Mason R. Eur Respir J 2020; in press (https://erj.ersjournals.com/content/55/4/2000607).
- Basic fibroblast growth factor protects against influenza A virus-induced acute lung injury by recruiting neutrophils. Wang K, Lai C, et. al. J Mol Cell Bio 2018; 10: 573–585. https://pubmed.ncbi.nlm.nih.gov/29121325/
- Receptor Specificity of the Fibroblast Growth Factor Family. The complete mammalian FGF family. Zhang X, Ibrahimi O, et al. J Biol Chem 2006; 281:15694 –15700. https://pubmed.ncbi.nlm.nih.gov/16597617/